MRP:
₹10
Find What You’re Looking For
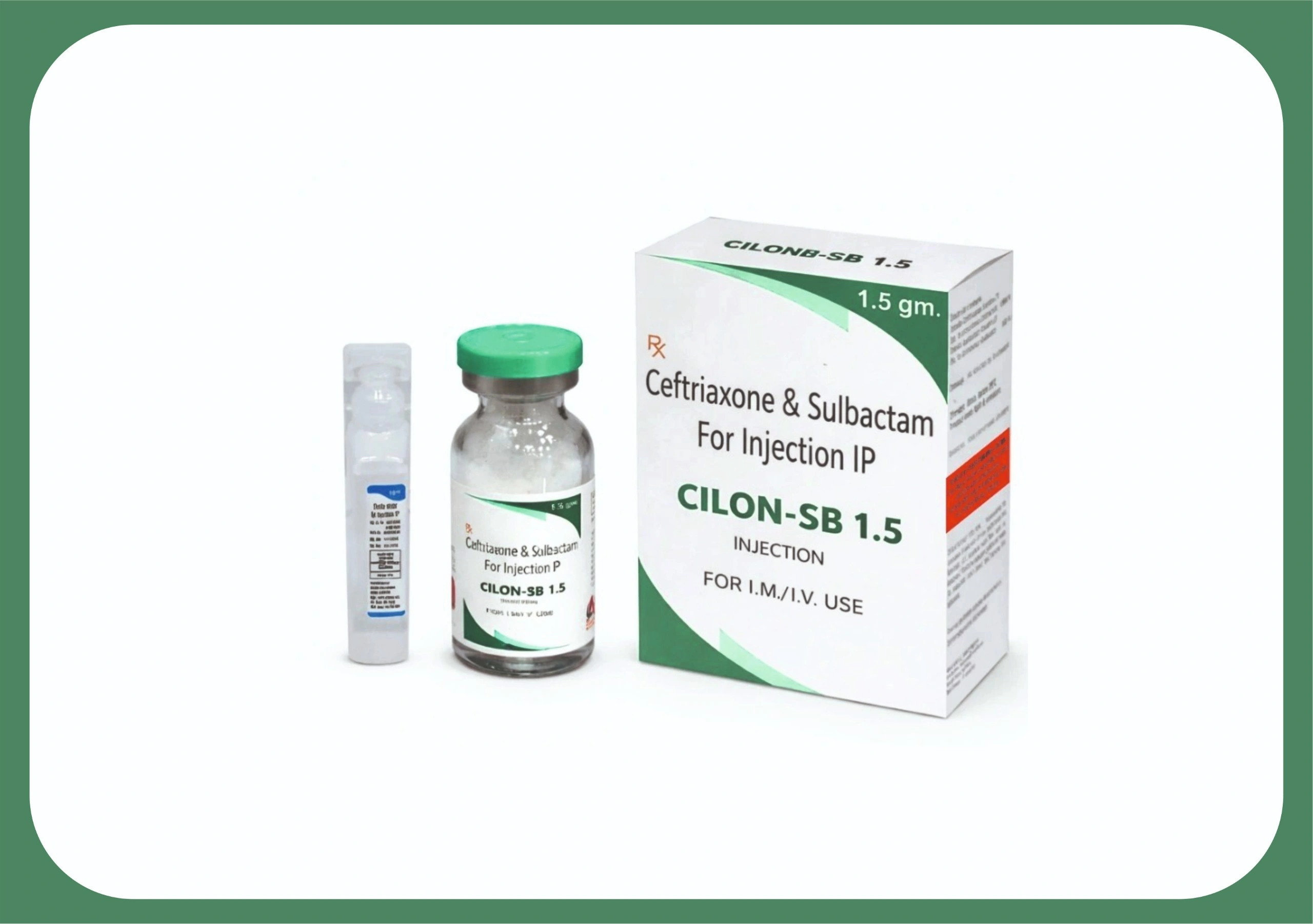

CILON SB 1.5 INJECTION
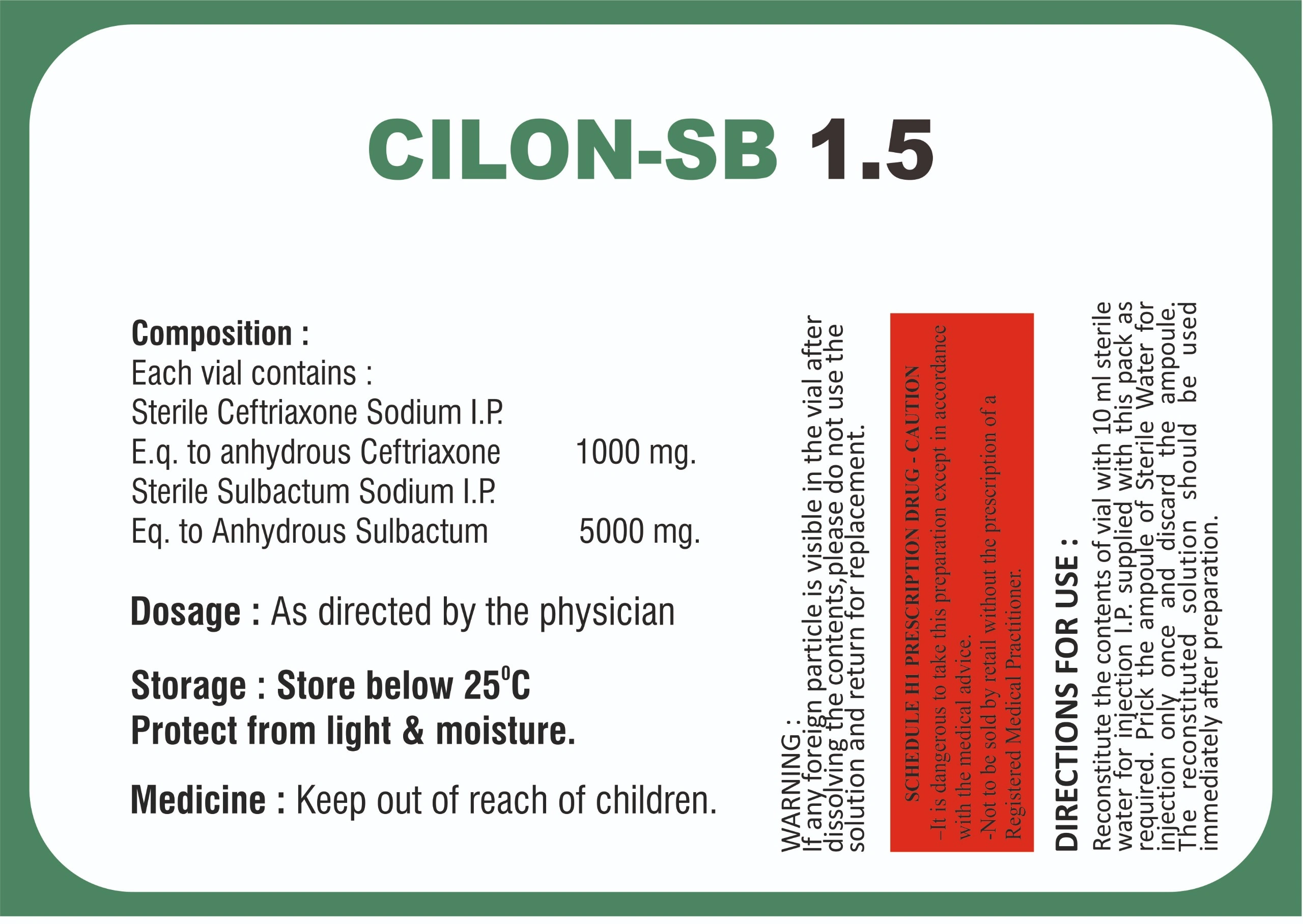
| COMPOSITION | CEFTRIAXONE 1000 MG + SULBACTUM 500 MG |
| MRP | 126.56 |
| PACKING SIZE | 1500 MG |
| PACKING TYPE | VIAL (WITH DISTILLED WATER) |
| FORM | INJECTION |
| GST | 5% |
| GRADE STANDARD | MEDICINE |
DESCRIPTION
Cilon SB 1.5 injection (Ceftriaxone 1000 mg + Sulbactam 500 mg) is a sterile injectable antibiotic formulation administered under medical supervision. It combines ceftriaxone, a third-generation cephalosporin, with sulbactam, a beta-lactamase inhibitor, to enhance antibacterial effectiveness against susceptible organisms. This combination helps in the treatment of moderate to severe bacterial infections by preventing bacterial resistance and improving overall therapeutic efficacy.
INDICATION
• Bacterial infections: Cilon SB 1.5 injection (Ceftriaxone 1000 mg + Sulbactam 500 mg) is prescribed for infections caused by susceptible bacteria. These include septicemia, pelvic inflammatory disease, intra-abdominal infections, urinary tract infections, and bone and joint infections.

• Bacterial meningitis: Cilon SB 1.5 injection (Ceftriaxone 1000 mg + Sulbactam 500 mg) is used in the treatment of meningitis caused by organisms such as Neisseria meningitidis and Streptococcus pneumoniae.
• Skin and soft tissue infections: Cilon SB 1.5 injection (Ceftriaxone 1000 mg + Sulbactam 500 mg) is effective against infections like cellulitis, abscesses, infected wounds, ulcers, and folliculitis caused by sensitive bacteria.

• Acute bacterial otitis media: Cilon SB 1.5 GM injection (Ceftriaxone 1000 mg + Sulbactam 500 mg) is used to treat middle ear infections caused by bacteria such as Haemophilus influenzae and Streptococcus pneumoniae.

• Lower respiratory tract infections: Cilon SB 1.5 injection (Ceftriaxone 1000 mg + Sulbactam 500 mg) is indicated for conditions like bacterial pneumonia.
• Surgical prophylaxis: Cilon SB 1.5 injection (Ceftriaxone 1000 mg + Sulbactam 500 mg) may be administered before surgery to reduce the risk of postoperative bacterial infections.
MECHANISM OF ACTION
• Ceftriaxone: This broad-spectrum antibiotic inhibits bacterial cell wall synthesis by binding to penicillin-binding proteins (PBPs), leading to weakening of the cell wall and bacterial cell death.
• Sulbactam: It inhibits beta-lactamase enzymes produced by certain bacteria, protecting ceftriaxone from degradation and extending its antibacterial coverage.
DRUG INTERACTIONS
Drug-Drug Intuitive: Cilon SB 1.5 injection (Ceftriaxone 1000 mg + Sulbactam 500 mg) may interact with cholera vaccine, ethinyl estradiol, mezlocillin, azlocillin, aminoglycosides (amikacin, streptomycin, neomycin, gentamicin), cyclosporine, and probenecid.
Drug-Food Intelligent: No intuitive found.
Intuitive Drug-Disease: Patients with kidney disease, liver disorders, gastrointestinal conditions, seizure disorders, diabetes, or bleeding disorders should use Cilon SB 1.5 injection (Ceftriaxone 1000 mg + Sulbactam 500 mg) with caution.
SIDE EFFECTS OF CILON SB 1.5 GM INJECTION
Major & minor side effects for Cilon SB 1.5 Injection
• Nausea
• Diarrhea
• Rash
• Injection site pain
PRECAUTIONS
Inform your healthcare provider if you are allergic to cephalosporins, penicillins, or other beta-lactam antibiotics due to possible cross-reactivity. Use cautiously in patients with kidney or liver impairment, gastrointestinal disorders, or bleeding conditions. Inform your doctor about all medications you are taking, including over-the-counter drugs. Monitor renal and hepatic function when required and avoid mixing with calcium-containing solutions, especially in neonates. Report any severe or unusual side effects promptly.
DISCLAIMER
The information on this website is provided for general informational purposes only and is not medical advice. It is not a substitute for professional medical advice, diagnosis, or treatment. Consult a qualified healthcare provider for personalized advice. We do not endorse any specific products, treatments, or services mentioned.
CILON SB 1.5 INJECTION



Speak with Us – Enquire Now!